Introduction
Genotyping plays an important role in the diagnosis, risk stratification and treatment response surveillance of lymphoma, a disease with considerable heterogeneity in clinical and pathological characteristics. To date, tumor tissue biopsy based genotyping is still considered as gold standard in lymphoma, but its application is hampered by the invasive nature and imperfect performance in capturing spatial and temporal tumor heterogeneity. Circulating tumor DNA (ctDNA), which is shed into peripheral blood by apoptotic or necrotic tumor cells, has shown the potential as a noninvasive alternative to tumor tissue DNA (tDNA) for molecular profiling. Many efforts have been made to investigate the feasibility of ctDNA based on next-generation sequencing (NGS) in lymphoma, but most of the studies were conducted in Caucasian population and limited to B-cell lymphoma patients. In the present study, we evaluated the performance of plasma-derived ctDNA against tDNA based genotyping across multiple lymphoma subtypes.
Methods
Between 4/2021 and 5/2023, a total of 110 patients were enrolled in this study. Matched tumor tissue biopsies and peripheral blood were collected at diagnosis. Both tDNA and ctDNA were extracted and subjected to a targeted capture NGS assay covering the entire coding region or recurrently mutated region of 196 genes. The mean coverage depth was 2431x and 2513x for tDNA and ctDNA, respectively. The limit of detection (LOD) was 1% and 0.5% for tDNA and ctDNA, respectively.
Results
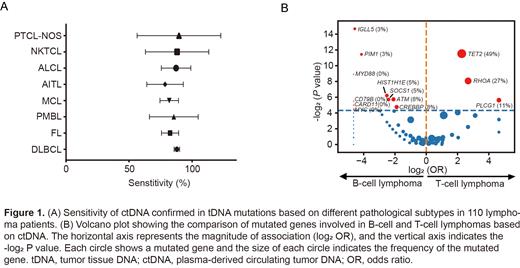
The enrolled cohort comprised of 63 (57.3%) male and 47(42.7%) female patients, with a median age of 58 years old (range 1.25-89). Among the 110 patients, the most prevalent subtype is diffuse large B-cell lymphoma (DLBCL,n=45), following by angioimmunoblastic T-cell lymphoma (AITL, n=16), anaplastic large cell lymphoma (ALCL, n=13) , mantle cell lymphoma (MCL, n=10), follicular lymphoma (FL, n=9), primary mediastinal large B-cell lymphoma (PMBL, n=9), extranodal natural killer/T cell lymphoma (ENKTL, n=4), and peripheral T-cell lymphoma not otherwise specified (PTCL-NOS, n=4). For further analysis, DLBCL, MCL, FL and PMBL were categorized as B-cell lymphoma group (n=73), while AITL, ALCL, ENKTL and PTCL-NOS were categorized as T-cell lymphoma group (n=37). Mutations were detected in 109(99.10%)tDNA and 110(100%)ctDNA, respectively. A total of 1586 mutations involving 177 genes were detected in tDNA, and the median number of mutations per sample was 12 (range 0-62), with KMT2D (39.09%), KMT2C (30.90%), IGLL5 (30.00%), TET2 (25.45%) and TP53 (20.90%) being the top 5 mutated genes. For ctDNA, a total of 1750 mutations involving 178 genes were detected and the median number of mutations per sample was 14 (range 2-63), with KMT2D (37.27%), KMT2C (29.09%), IGLL5 (27.27%), TET2 (25.45%) and DNMT3A (27.27%) being the top 5 mutated genes. To determine the concordance of mutations detected in tDNA and ctDNA, sensitivity was calculated utilizing tDNA as gold standard. The overall sensitivity of ctDNA based genotyping was 85.39% (95%CI: 82.36%-88.41%), with B-cell lymphoma showing a slightly higher sensitivity than T-cell lymphoma (86.17%, 95%CI: 83.51%-88.83% vs. 83.84%, 95%CI: 76.30%-91.32%). In terms of subtypes, PTCL-NOS exhibited the highest sensitivity (89.58%, 95%CI: 56.43%-122.73%, while AITL exhibited the lowest sensitivity (78.53%, 95%CI: 64.09%-92.97%) (Figure 1A). In addition, the difference of mutational spectrum between B-cell lymphoma and T-cell lymphoma was precisely depicted by ctDNA based genotyping. We discovered mutations in TET2, RHOA, and PLCG1 were significantly present in T-cell lymphoma, while mutations in MYD88, CD79B, CARD11, CREBBP, MYC, PIM1, IGLL5, ATM, SOCS1 and HIST1HIE were significantly enriched in B-cell lymphoma (Figure 1B).
Conclusion
In the present study, recurrent mutations across a variety of lymphoma subtypes were identified by ctDNA based genotyping, which were in high concordance with tDNA based genotyping. These data demonstrated that ctDNA based genotyping is a reliable noninvasive alternative to tumor tissue biopsy for molecular profiling of lymphoma patients, especially for patients whose tissues are not accessible for genotyping.
Disclosures
Yuan:AcornMed Biotechnology Co., Ltd.: Current Employment. Wang:AcornMed Biotechnology Co., Ltd.: Current Employment. Zou:AcornMed Biotechnology Co., Ltd.: Current Employment.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal